子宮頸がん
子宮がんはよく耳にすることが多いと思いますが、子宮がんには子宮頸がんと子宮がんの2つがあり、これらの違いをご存知ですか?これらのがんは原因も多発する年齢層も全く異なります。 ここではこの2つのが ...

子宮がんはよく耳にすることが多いと思いますが、子宮がんには子宮頸がんと子宮がんの2つがあり、これらの違いをご存知ですか?これらのがんは原因も多発する年齢層も全く異なります。 ここではこの2つのが ...

がんの原因の約5%が遺伝的要因と言われています。 このことを家族性腫瘍と呼びます。 ですが、がんの原因は遺伝よりも、喫煙や野菜不足などの生活習慣の影響の方が多いと言われ、これらの理由は全体の約6 ...
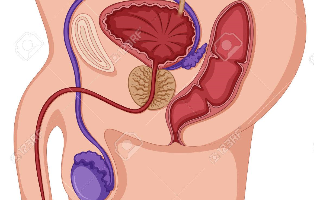
前立腺がんは罹患率の高いものの生存率は比較的高いがんです。日本において前立腺がんの罹患率は、胃がん、肺がん、大腸に次いで第4位で、死亡率は男性人口10万人あたり18.9人です。死亡率が肺がんでは86人 ...

がんの標準治療は、3大治療法と呼ばれる手術療法・放射線療法・抗がん剤療法です。その中でも特に手術療法でがん腫瘍を全て切り取ることが、一番の根治治療とされてきました。 がん細胞が局所的に存在する場合は ...

2人に1人はがんにかかると言われる昨今。決して珍しいがんではありません。血縁者にたとえがんになった方が複数いても、遺伝性のがんとは言えません。特に大腸がんの場合、遺伝するケースは非常に稀と言われており ...

がんの骨転移が起こる状態は、がんが進行していると考えられますが、早期発見し治療を早く開始する事で、共存共生が望めるものなのです。 他の内臓への転移と違い骨転移が原因で命を落とすような事は有りません。 ...
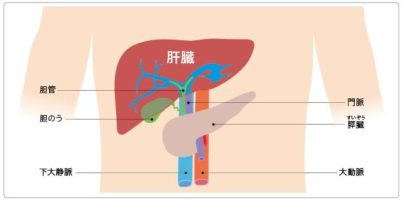
肝臓は機能の低下があっても滅多に症状が現れない、「沈黙の臓器」と言われています。 体内において数多くの機能をこなしている肝臓が正常に働かなくなると、私たちは生命を維持していくことができません。 そ ...

大腸ポリープには良性のものと悪性のものがあります。健康診断でポリープや腫瘍が見つかると、がんではないかと心配になるかもしれません。大腸ポリープの8割は腺腫で、特に直腸、S状結腸によくできます。大腸がん ...
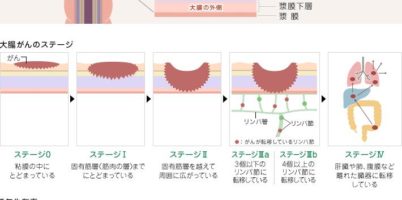
大腸がんは日本において、罹患率の高いがんとして知られていますが、早期発見できれば、完治する確率も高いがんとしても知られています。近年、日本における患者数の増加が多い事から、生活環境の変化に着眼し、食の ...

罹患率の高い前立腺がんですが、一方で生存率は高いがんです。日本において前立腺がんの罹患率は、男性人口10万人あたり117.9人で、胃がん、肺がん、大腸に次いで第4位でが、死亡率は男性人口10万人あたり ...