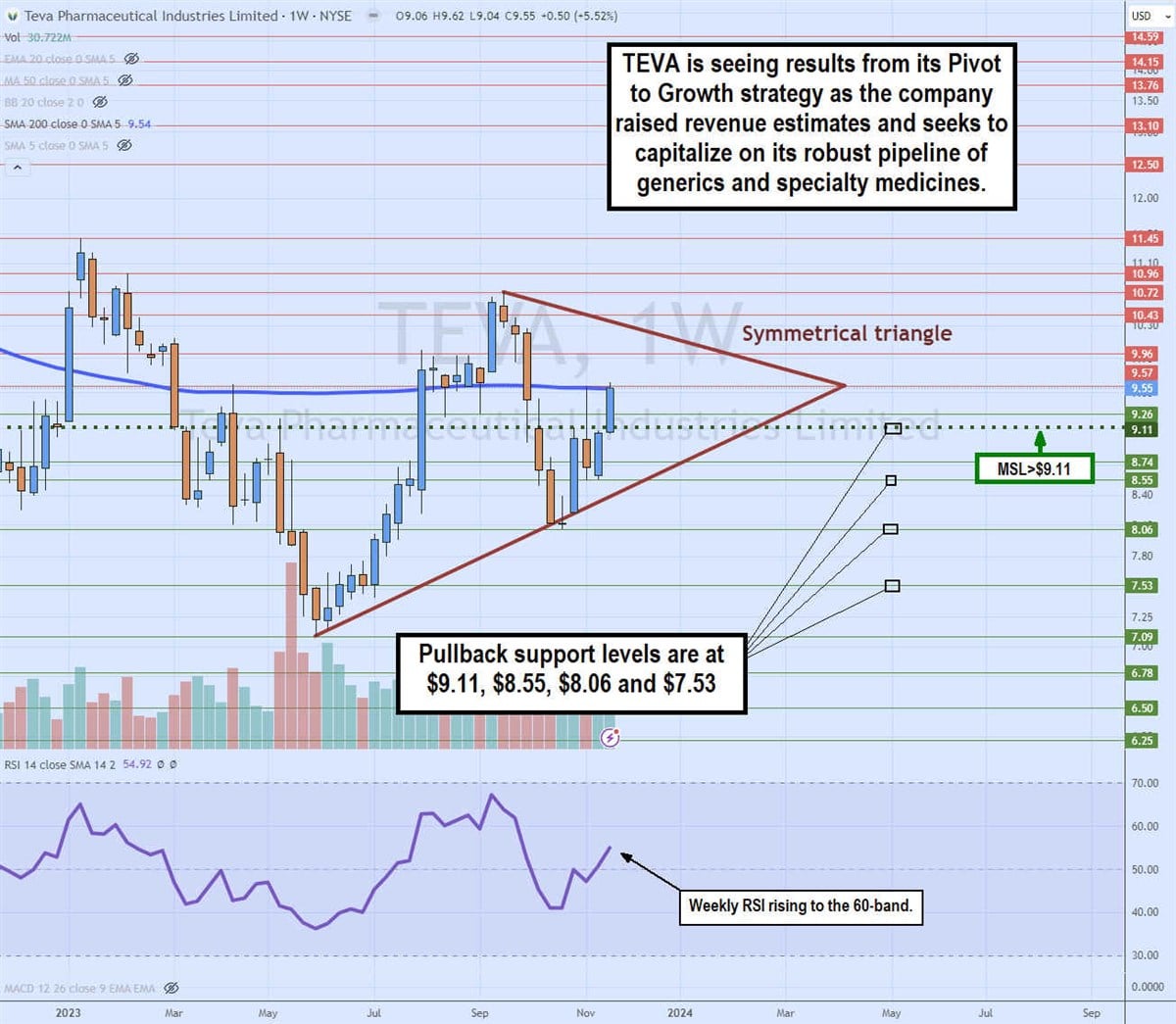
Teva Announces Approval of a Generic Version of Forteo® (teriparatide injection), in the U.S. | Business Wire

Teva Announces Approval of a Generic Version of Forteo® (teriparatide injection), in the U.S. | TEVA Stock News
Teva Announces Approval of a Generic Version of Forteo® (teriparatide injection), in the U.S. -November 17, 2023 at 08:31 am EST | MarketScreener
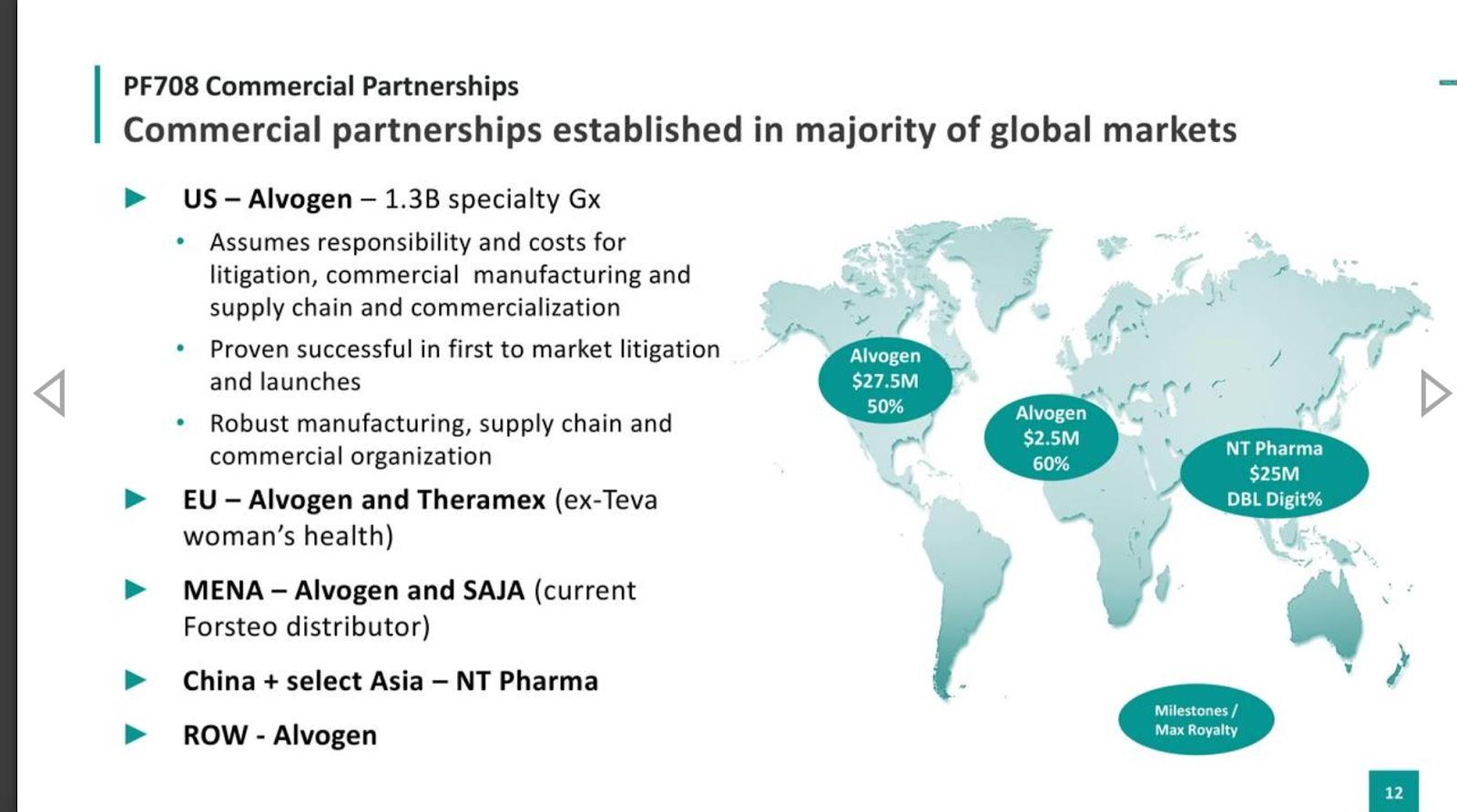
Pfenex: The First U.S. Forteo Biosim And A Host Of Other Biosims In Pipeline (NYSEMKT:PFNX) | Seeking Alpha

How the U.S. Compares to Europe on Biosimilar Approvals and Products In the Pipeline | Biosimilars Law Bulletin

How the U.S. Compares to Europe on Biosimilar Approvals and Products In the Pipeline Updated March 14, 2022 | Rothwell, Figg, Ernst & Manbeck, P.C. - JDSupra

How the U.S. Compares to Europe on Biosimilar Approvals and Products In the Pipeline Updated March 14, 2022 | Biosimilars Law Bulletin